News

March 10. - 2026
Biologging Reveals Hidden Effects of Sedative Drugs
A recent study published in Proceedings of the National Academy of Sciences (PNAS) sheds new light on how the brain regulates body temperature during sedation. The research focused on the locus coeruleus, a small region of the brain known for producing norepinephrine and playing a central role in arousal and sleep regulation. Scientists investigated how this neural circuit influences thermoregulation during sedation with dexmedetomidine, a drug widely used in intensive care because it produces a sleep-like state while allowing patients to be easily awakened.
What Drives DEX-Induced Hypothermia?
While hypothermia can be beneficial in some instances it can be detrimental in others, especially when induced inadvertently. Norepinephrine’s is usually associated with arousal; however, it’s regulation in sleep is more complex. Research shows that the sedative α2‑agonist dexmedetomidine (DEX) can selectively activate a branch of the norepinephrine (NE) system to induce non-rapid eye movement (NREM)‑like sleep, marked by strong delta‑wave activity and reduced REM sleep. DEX also triggers torpor‑like hypothermia, lowering body temperature, heart rate, and metabolism effects that are clinically relevant given the risks of inadvertent hypothermia during sedation. New findings reveal that while the locus coeruleus (LC) modulates sensitivity to DEX, it is not required for DEX‑induced NREM‑like sleep or hypothermia, instead, adra2a receptors in the medial preoptic hypothalamus play a key role. These insights deepen our understanding of how sedatives interact with thermoregulation and sleep circuits, with implications for anesthesia, critical care, and neurodegenerative disease risk.
Mice with ablated LC showed lower body temperature compared to the control group
Scientists from Imperial College, UK, University of Zurich and ETH Zurich, Switzerland implanted mice with Star-Oddi’s DST nano-T temperature loggers. The loggers measured core body temperature in control mice as well as mice with lesioned LC (ΔLC mice), while comparing the effects of different dosages of DEX on sleep and body temperature. The effects of DEX under various conditions were then explored. Before DEX administration the ΔLC mice show normal sleep–wake patterns but have consistently lower core body temperatures, about 1 °C cooler during lights‑off and 0.5 °C cooler during lights‑on, with reduced daily max and min temperature compared to the control group.
What Drives DEX-Induced Hypothermia?
While hypothermia can be beneficial in some instances it can be detrimental in others, especially when induced inadvertently. Norepinephrine’s is usually associated with arousal; however, it’s regulation in sleep is more complex. Research shows that the sedative α2‑agonist dexmedetomidine (DEX) can selectively activate a branch of the norepinephrine (NE) system to induce non-rapid eye movement (NREM)‑like sleep, marked by strong delta‑wave activity and reduced REM sleep. DEX also triggers torpor‑like hypothermia, lowering body temperature, heart rate, and metabolism effects that are clinically relevant given the risks of inadvertent hypothermia during sedation. New findings reveal that while the locus coeruleus (LC) modulates sensitivity to DEX, it is not required for DEX‑induced NREM‑like sleep or hypothermia, instead, adra2a receptors in the medial preoptic hypothalamus play a key role. These insights deepen our understanding of how sedatives interact with thermoregulation and sleep circuits, with implications for anesthesia, critical care, and neurodegenerative disease risk.
Mice with ablated LC showed lower body temperature compared to the control group
Scientists from Imperial College, UK, University of Zurich and ETH Zurich, Switzerland implanted mice with Star-Oddi’s DST nano-T temperature loggers. The loggers measured core body temperature in control mice as well as mice with lesioned LC (ΔLC mice), while comparing the effects of different dosages of DEX on sleep and body temperature. The effects of DEX under various conditions were then explored. Before DEX administration the ΔLC mice show normal sleep–wake patterns but have consistently lower core body temperatures, about 1 °C cooler during lights‑off and 0.5 °C cooler during lights‑on, with reduced daily max and min temperature compared to the control group.
External warming can prevent mortality of DEX induced hypothermia
DEX causes dramatically stronger and longer‑lasting hypothermia in ΔLC mice, with mild cooling in controls, but up to a 10 °C drop for ~6 h in ΔLC animals, escalating with dose to the point that 200 µg/kg leads to sustained 24‑h hypothermia and 100% mortality (se fig 2. below).
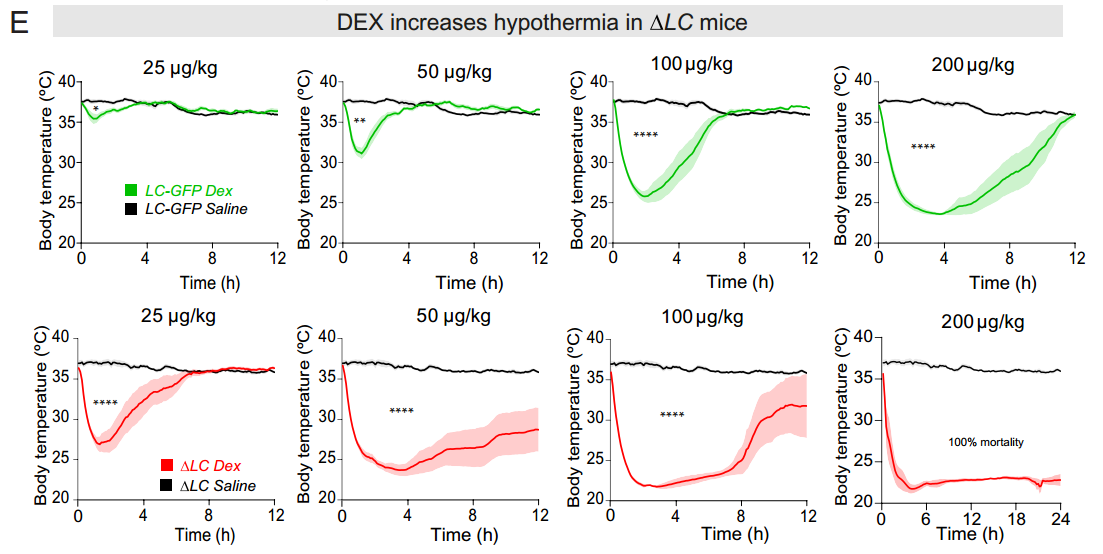
From fig. 2 showing body temperature in the control group vs. ΔLC, receiving different doses of DEX.
External warming at 32 °C prevents the extreme DEX‑induced hypothermia in ΔLC mice, making even the previously lethal 200 µg/kg dose survivable, and allows their temperature and NREM‑like sleep responses to resemble the sleep in the control group, showing that the LC is unnecessary for DEX‑induced sedation and that the exaggerated sleep seen at 21 °C was driven by hypothermia.
DEX‑induced hypothermia is strongly reduced and recovers faster when adra2a receptors are knocked down in MPO/septum Vglut2 glutamate neurons (fig. 5 below), showing that these cells primarily drive DEX’s temperature‑lowering effects rather than its NREM‑like sleep.
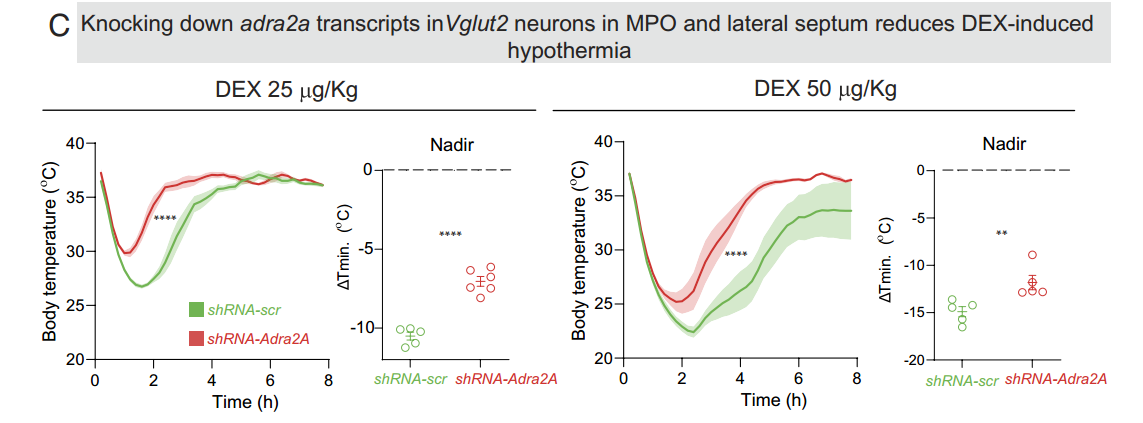
From fig. 5 showing reduction in DEX-induced hypothermia by knocking down adra2a receptors.
LC‑independent hypothalamic circuitry drives DEX‑induced hypothermia and NREM‑Like sleep
DEX’s actions on sleep and thermoregulation arise from a distributed hypothalamic circuit rather than direct inhibition of the locus coeruleus. Loss of LC‑derived norepinephrine upregulates adra2a receptors in MPO and forebrain neurons, making ΔLC mice profoundly hypersensitive to DEX‑induced hypothermia and NREM‑like sleep. External warming normalizes these exaggerated responses, revealing that the LC functions mainly as a thermoregulatory brake that speeds recovery from hypothermia rather than as DEX’s primary sedative target. These findings refine the textbook model of α2‑adrenergic sedation, highlight the importance of temperature management during DEX use, and suggest that individuals with LC degeneration, such as in early Alzheimer’s disease, may be more vulnerable to hypothermic complications under anesthesia.
Further results can be view in the article itself published in PNAS and can be accessed here.


